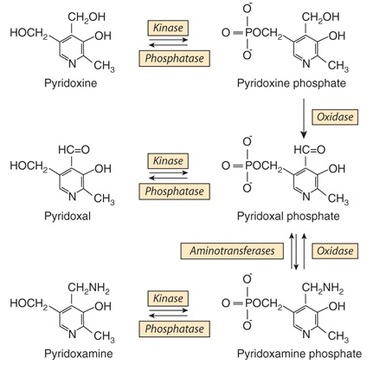
Six compounds have vitamin B6 activity (Figure 12): pyridoxine, pyridoxal, pyridoxamine, and their 5'phosphates. The active coenzyme is pyridoxal 5'-phosphate. Some 80% of the body's total vitamin B is pyridoxal phosphate in muscle, mostly associated with glycogen phosphorylase. This is not available in deficiency, but is released in starvation, when glycogen reserves become depleted, and is then available, especially in liver and kidney, to meet increased requirement for gluconeogenesis from amino acids (Murray et. al. 2009).
|
Pyridoxal phosphate is a coenzyme for many enzymes involved in amino acid metabolism, especially transamination and decarboxylation. It is also the cofactor of glycogen phosphorylase, where the phosphate group is catalytically important. In addition, B6 is important in steroid hormone action. Pyridoxal phosphate removes the hormone-receptor complex from DNA binding, terminating the action of the hormones. In vitamin B6 deficiency, there is increased sensitivity to the actions of low concentrations of estrogens, androgens, cortisol, and vitamin D (Murray et. al. 2009).
Although clinical deficiency disease is rare, there is evidence that a significant proportion of the population have marginal |
vitamin B6 status. Moderate deficiency results in abnormalities of tryptophan and methionine metabolism. Increased sensitivity to steroid hormone action may be important in the development of hormone-dependent cancer of the breast, uterus, and prostate, and vitamin B6 (Murray et. al. 2009).