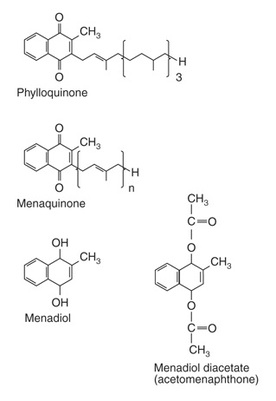
Vitamin K, which exist in several forms, contains aromatic rings with side chains of varying length. The different forms of Vitamin K are (Figure 5); Phylloquinone (Vit. K1) - present in green vegetables, the normal dietary source; Menaquinone (Vit. K2) - synthesized by intestinal bacteria with differing lengths of side chain; Menadiol, Menadione (Vit. K3), Menadiolo Acetate – synthetic derivatives which can be metabolized to phylloquinone. (Figure _.)
Vitamin K is the cofactor for the carboxylation of glutamate residues in the post-synthetic modification of proteins to form the unusual amino acid γ-carboxyglutamate (Gla). Initially, Vitamin K hydroquinone is oxidized to the epoxide, which activates a glutamate residue in the protein substrate to a carbanion, which reacts nonenzymically with carbon dioxide to form γ-carboxyglutamate. Vitamin K epoxide is reduced to the quinone by a warfarin-sensitive reductase, and the quinone is reduced to the active hydroquinone by either the same warfarin sensitive reductase or a warfarin-insensitive quinone reductase. In the presence of warfarin, vitamin K epoxide cannot be reduced, but accumulates and is excreted. If enough vitamin K (as the quinone) is provided in the diet, it can be reduced to the active hydroquinone by the warfarin-insensitive enzyme, and carboxylation can continue, with stoichiometric utilization of vitamin K and excretion of the epoxide (Murray et. al. 2009).
Prothrombin and several other proteins of the blood clotting system (Factors VII, IX, and X, and proteins C and S, Chapter 50) each contain 4–6 γ-carboxyglutamate residues. γ-Carboxyglutamate chelates calcium ions, and so permits the binding of the blood clotting proteins to membranes. In vitamin K deficiency, or in the presence of warfarin, an abnormal precursor of prothrombin (preprothrombin) containing little or no -carboxyglutamate, and incapable of chelating calcium, is released into the circulation (Murray et. al. 2009).
Prothrombin and several other proteins of the blood clotting system (Factors VII, IX, and X, and proteins C and S, Chapter 50) each contain 4–6 γ-carboxyglutamate residues. γ-Carboxyglutamate chelates calcium ions, and so permits the binding of the blood clotting proteins to membranes. In vitamin K deficiency, or in the presence of warfarin, an abnormal precursor of prothrombin (preprothrombin) containing little or no -carboxyglutamate, and incapable of chelating calcium, is released into the circulation (Murray et. al. 2009).