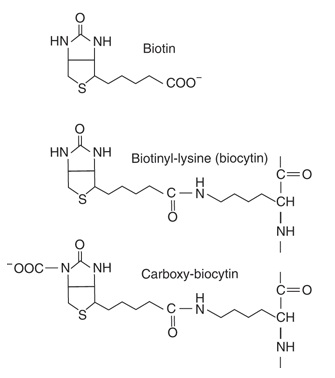
The structures of biotin, biocytin, and carboxybiotin (the active metabolic intermediate) are shown in Figure 13. Biotin is widely distributed in many foods as biocytin (ε-amino-biotinyllysine), which is released on proteolysis. It is synthesized by intestinal flora in excess of requirements. Deficiency is unknown, except among people maintained for many months on total parenteral nutrition, and a very small number who eat abnormally large amounts of uncooked egg white, which contains avidin, a protein that binds biotin and renders it unavailable for absorption (Murray et. al. 2009).
|
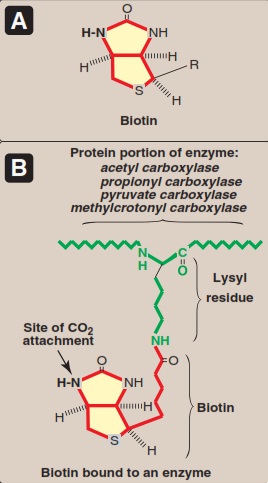
Biotin (Figure 14) functions to transfer carbon dioxide in a small number of reactions: acetyl-CoA carboxylase, pyruvate carboxylase , propionyl-CoA carboxylase, and methylcrotonyl-CoA carboxylase. A holocarboxylase synthetase catalyzes the transfer of biotin onto a lysine residue of the apo-enzyme to form the biocytin residue of the holoenzyme. The reactive intermediate is 1-N -carboxy-biocytin, formed from bicarbonate in an ATP-dependent reaction. The carboxy group is then transferred to the substrate for carboxylation. Biotin also has a role in regulation of the cell cycle, acting to biotinylate key nuclear proteins (Murray et. al. 2009).
|